- Welcome to KIDLAB!
- About the Teacher
- Classes
- Announcements
- Contact Ms. O
- Science Events in Boston
- Fun Science Sites for kids!
- Donations
- Q & A
- Guest Book
- Archives
- Super STEM Saturday!>
- 2009-2010 KIDLAB>
- 2010-2011 KIDLAB>
- 2011-2012 KIDLAB>
- 2012-2013 KIDLAB>
- K2- Fall Exploration
- K2- Float or Sink?
- K2 Boats
- boat images
- G1 Super Science Centers
- G1 Wind!
- G1 Living Museum Props
- G1 A Reason for the Seasons
- Caterpillar images
- G2 Super Science Centers
- G2 Fantastic Forests
- G2 Dinosaurs are DINOmite!
- Dinosaur pictures
- G3 Earthbound
- G3 Blast Off!
- G3 Stargazing
- G3/G4 Welcome to the NHCS Zoo!
- G4 Wonderful Water
- G4 Clean It Like Mother Nature!
- G4 Adam, the Atom
- Maps of the United States
- Cloud Pictures
- Anatomy of a Flower
- G5 Lift it with a Lever!
- G5 Pinball Wizard
- G5 Rube Goldberg Machines
- Images of lunar landing space crafts
- G6/7 Biomimicry and Arthropods
- Enrichment- Shadow Boxes
- Famous buildings
- Types of Plants
Water, Water, Everywhere
In science class with Ms. Baugher, the 4th grade has been learning about the water cycle. Water is one of the most abundant substances and earth and is vital to survival but what exactly is water and what makes it change?
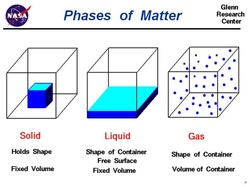
In KIDLAB, students looked at water on the molecular level. We discussed that all things are made out of atoms and when atoms combine, they make molecules. Water is a molecule consisting of two atoms of hydrogen and one atom of oxygen, hence H2O. Sometimes molecules of water are close together and form a solid (ice) while sometimes they are far away from each other and are a gas (water vapor). Water as a liquid has molecular movement in the middle with molecules not super close together but far enough away from each other that they can slide past. It is for this reason that liquids can pour and move while solids keep their shape. Check out the projects we created to keep these ideas fresh in our minds!
In KIDLAB, students looked at water on the molecular level. We discussed that all things are made out of atoms and when atoms combine, they make molecules. Water is a molecule consisting of two atoms of hydrogen and one atom of oxygen, hence H2O. Sometimes molecules of water are close together and form a solid (ice) while sometimes they are far away from each other and are a gas (water vapor). Water as a liquid has molecular movement in the middle with molecules not super close together but far enough away from each other that they can slide past. It is for this reason that liquids can pour and move while solids keep their shape. Check out the projects we created to keep these ideas fresh in our minds!
